Ultrasound-Facilitated Endovascular System for Acute PE shown to Lower Pulmonary Hypertension
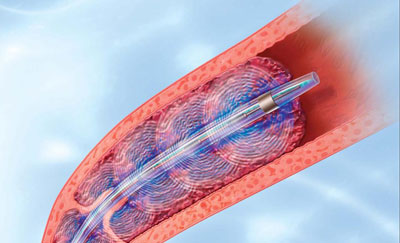
 Gregory Piazza, MD, clinical instructor in medicine at Harvard Medical School, presented the results of a single-arm, multi center trial that tested the efficacy of the EkoSonic endovascular system. 150 patients, all with proximal pulmonary embolism (PE) and a dilated right ventricle (RV/VL ratio ≥0.9) were examined using ultrasound-delivered, catheter-directed low-dose thrombolysis. Thirty-one patients had acute massive PE and 119 had sub-massive PE.
Gregory Piazza, MD, clinical instructor in medicine at Harvard Medical School, presented the results of a single-arm, multi center trial that tested the efficacy of the EkoSonic endovascular system. 150 patients, all with proximal pulmonary embolism (PE) and a dilated right ventricle (RV/VL ratio ≥0.9) were examined using ultrasound-delivered, catheter-directed low-dose thrombolysis. Thirty-one patients had acute massive PE and 119 had sub-massive PE.
24 mg of tissue plasmogen activator (tPA) was administered either as 1 mg/hr for 24 hours with a unilateral catheter or 1 mg/hour/catheter for 12 hours with bilateral catheters. Researchers report that the Ekos EkoSonic endovascular system improved right ventricle function and demonstrated a decrease in pulmonary hypertension and intracranial hemorrhage risk.
The SEATTLE II study, according to a press release, reported that the mean RV/LV ratio decreased significantly from 1.55 before thrombolysis to 1.13 at 48 hours after tPA administration (P<.0001). No deaths were reported among patients with massive PE at a thirty-day follow. Overall, one death occurred that was attributed to PE.
Researchers report that there were no fatal bleeding events or intracranial hemorrhage, however 17 major bleeding events were reported in 17 patients which included 16 moderate bleeds and one severe. Six of the major bleeds occurred in patients with comorbidities known to be linked with increased bleeding risk during thrombolytic therapy.
Samuel Z. Goldhaber, MD, principal investigator and professor of medicine at Harvard Medical School and director of the Thrombosis Research Group at Brigham and Women’s Hospital, said in a press release, “This trial represents a breakthrough in demonstrating the safety and efficacy of thrombolytic therapy for acute PE. The reduction of the RV/LV ratio by 0.42 is substantial and clinically significant, without any intracranial hemorrhage and using a much-reduced lytic dose. These findings establish a new rationale for considering thrombolysis in both massive and sub-massive PE.”
In the United States, the EkoSonic endovascular system has been indicated for controlled and selective infusion of physician-specific fluids, including thrombolytics, in the peripheral blood vessels. However, in Europe, the system is intended for PE therapy on patients with ≥50% clot burden in one or both main pulmonary arteries or lobar pulmonary arteries, and evidence of right ventricle dysfunction.
[adrotate group=”3″]