[vc_row][vc_column][vc_column_text]PRX-08066, also known as Maleic acid, is a novel, experimental, selective 5-hydroxytryptamine receptor 2B (5-HT2BR) antagonist targeted at treating patients with Pulmonary Arterial Hypertension (PAH) in order to improve exercise ability and reduce clinical disease progression. Currently under development by Epix Pharmaceuticals Inc., the drug is chemically known as 5-((4-(6-Chlorothieno [2, 3 -d] pyrimidin-4-ylamino) piperidin-1-yl) methyl)-2-fluorobenzonitrile monofumarate and is a selective vasodilator. To date, most of the PAH related therapies have focused on three main pathways, namely, endothelin, prostacyclin and nitric oxide, to mediate vasodilation. This drug is a novel formulation that targets the serotonin pathway to prevent vasoconstriction.
This treatment is no longer under development.
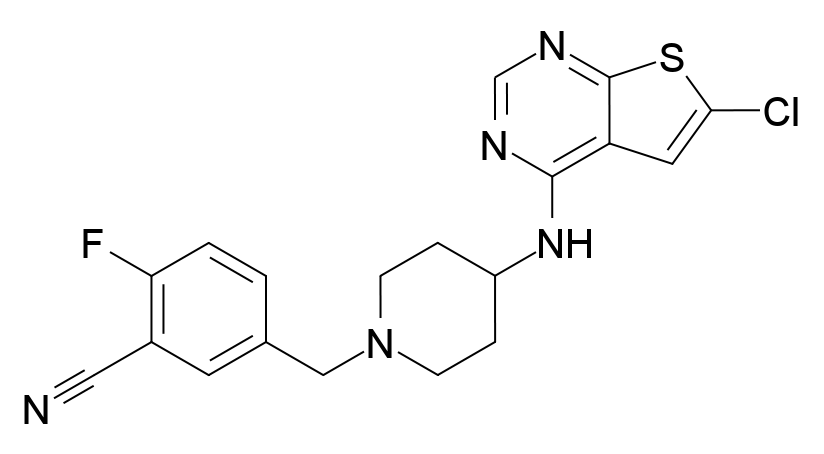 How PRX-08066 Works
How PRX-08066 Works
Serotonin (5-HT) is a potent pulmonary vasoconstrictor, mitogen and stimulator of migration of pulmonary artery smooth muscle cells. Previous research has shown that 5-HT plays a stimulatory role in vasoconstriction, promotion of platelet aggregation and pulmonary arterial smooth muscle proliferation as seen in pulmonary hypertension. 5-HT exerts its effects through various 5-HT receptors and a 5-HT transporter. Researchers have observed that the 5-HT 2B receptor’s (5-HT2BR) expression has been found to be elevated in patients with PAH (both in preclinical and clinical studies with human hosts), suggesting its pivotal role in the disease. 5-HT2BR is a kind of G-protein Coupled Receptor (GCR) that exists in abundance in the pulmonary endothelial and smooth muscle cells of humans and stimulate the release of calcium ions from the pulmonary arteries, along with induction of hypoxic responses within the pulmonary arteries, leading to stress build-up and eventually PAH. As a result, selective inhibition of these receptors could provide a way to reduce the right ventricular stress build-up and eventually cause vasodilation.
Studies Involving PRX-08066
Preclinical studies with PRX-08066 have shown that it possessed the ability to inhibit 5-HT-induced mitogen-activated protein kinase activation, which leads to excess calcium ion secretion and vasoconstriction. Moreover, it has also been observed to reduce hypoxia-induced right ventricular systolic pressure in mice and rat models, suggesting its role in selective vasodilation of pulmonary vasculature without causing any direct damage to systemic blood pressure. Human phase I and II clinical trials (data received as of March 2009), which followed these preclinical studies, were randomized, double-blind, placebo-controlled studies to evaluate safety profiles and efficacy of PRX-08066 in patients with PAH and COPD. The primary end-points of these studies were measured in terms of reduction in Systolic Pulmonary Arterial Pressure (SPAP) in comparison to the regular 6MWD tests in case of other drugs. The studies successfully showcased the drug’s ability to reduce SPAP and improve pulmonary and cardiac health, along with a good tolerance profile among patients. Further large scale phase III studies await the drug’s approval by the US FDA and subsequent commercialization.
This treatment is no longer in development
Note: Pulmonary Hypertension News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
Sources:
https://www.wikinvest.com/stock/EPIX_Pharmaceuticals_(EPIX)/Prx-08066_Pulmonary_Hypertension
https://clinicaltrials.gov/show/NCT00345774
https://www.phaonlineuniv.org/ResourceLibrary/Resource.cfm?ItemNumber=3852
https://jpet.aspetjournals.org/content/334/2/364.full
[/vc_column_text][/vc_column][/vc_row][vc_row][vc_column width=”1/2″][vc_wp_rss items=”7″ title=”Read the Latest News About Pulmonary Hypertension” url=”https://pulmonaryhypertensionnews.com/tag/pulmonary-hypertension/feed/”][/vc_column][vc_column width=”1/2″][vc_wp_text title=”Find Out More”]
[/vc_wp_text][/vc_column][/vc_row]
