Uptravi (selexipag) for pulmonary arterial hypertension
What is Uptravi for pulmonary arterial hypertension?
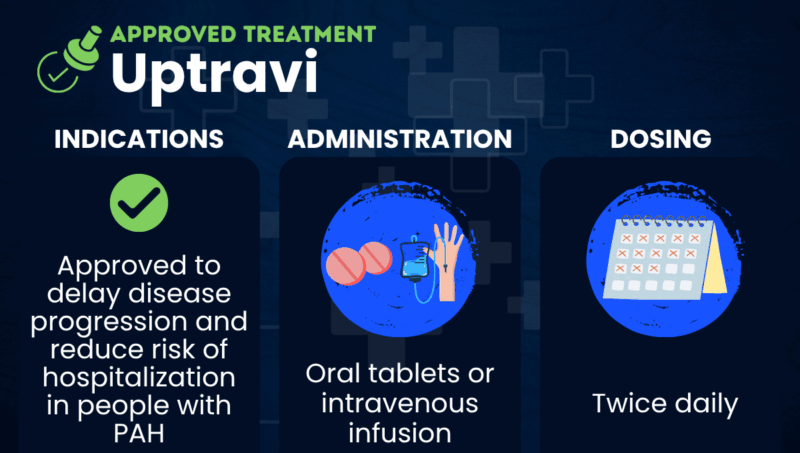
Uptravi (selexipag) is an approved therapy from Johnson & Johnson that’s used to delay disease progression and reduce the risk of hospitalization in people with pulmonary arterial hypertension (PAH). It is available in oral and intravenous (into-the-vein) formulations.
PAH occurs when the blood vessels in the lungs narrow and the pressure in them rises, making the heart work harder to pump blood through them. Over time, strain on the right side of the heart can lead to complications and more frequent hospital stays.
Uptravi is a prostacyclin receptor agonist that is designed to mimic the effects of prostacyclin, a naturally occurring molecule that helps blood vessels relax and widen to make blood flow easier. By binding to the same receptor proteins through which prostacyclin exerts these effects, Uptravi reduces blood pressure in the lungs and eases disease symptoms. Unlike some older prostacyclin therapies, Uptravi is designed to avoid activating prostacyclin receptors that could cause side effects.
Therapy snapshot
| Brand name | Uptravi |
| Chemical name | Selexipag |
| Usage | Used to delay disease progression and reduce the risk of hospitalization in people with PAH |
| Administration | Oral tablets or intravenous infusion |
Who can take Uptravi?
Uptravi is approved in the U.S. to delay disease progression and reduce the risk of hospitalization in people with PAH (World Health Organization Group 1). Its safety and effectiveness in children have not been established. Uptravi is similarly approved in other areas, including the European Union and Canada.
The medication is contraindicated, or should not be used, in patients who are:
- taking medications that strongly inhibit the CYP2C8 enzyme, as this can affect exposure to Uptravi in the body
- have a history of severe immune reactions to selexipag or any other ingredients in Uptravi.
How is Uptravi administered?
Uptravi is taken twice per day, usually in the form of oral tablets. The tablets can be taken with or without food, but food may help with tolerability issues.
The recommended starting dose is 200 micrograms (mcg), twice daily. It will then be increased at weekly intervals to the highest tolerated dose up to 1,600 mcg twice daily. The dosing regimen may need to be adjusted for people with liver impairment or who are taking certain medications.
Intravenous infusions of Uptravi can be given to patients who are temporarily unable to take the oral tablets. The twice-daily infusions are administered by a healthcare provider, with the dose based on the patient’s current oral dose. Each infusion takes 80 minutes.
Uptravi in clinical trials
The approval of Uptravi for PAH was based on data from GRIPHON (NCT01106014), a Phase 3 clinical trial that enrolled 1,156 adults with PAH, most of whom were already receiving standard PAH therapies. Participants were randomly assigned to receive either oral Uptravi or a placebo for about six months. Data showed:
- Treatment with Uptravi resulted in a 40% reduction in the risk of death or disease progression compared with the placebo. That difference was mainly attributed to fewer disease progression events and PAH-related hospitalizations with Uptravi.
- Uptravi was effective in a broad patient population, regardless of PAH cause, disease severity, or other treatments being used.
Data from GRIPHON’s open-label extension study (NCT01112306) also showed the therapy continued to be safe long term. Survival rates for those still in the study were 74% after five years and 60% after 10 years.
Another Phase 3 trial (NCT03187678) demonstrated the safety and tolerability of temporarily switching between oral and intravenous Uptravi.
Uptravi side effects
The most common side effects reported with Uptravi include:
- headache
- diarrhea
- jaw pain
- nausea
- muscle pain
- vomiting
- pain in the extremities
- flushing.
If fluid buildup in the lungs happens to a person using Uptravi, they should be evaluated for pulmonary veno-occlusive disease, a rare cause of PAH where the small veins in the lungs become blocked. If this blockage is confirmed, Uptravi should be discontinued.
Pulmonary Hypertension News is strictly a news and information website about the disease. It does not provide medical advice, diagnosis, or treatment. This content is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
 Fact-checked by
Fact-checked by 


