A New Molecular Mechanism for Pulmonary Hypertension That Can Be Corrected
Written by |
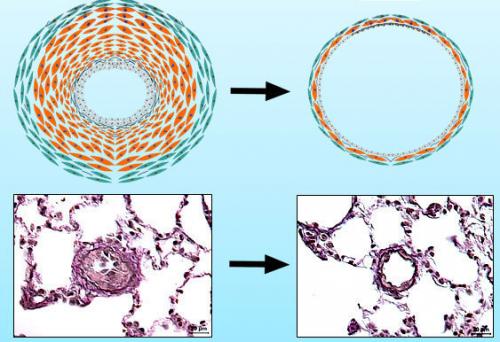
 At the Max Planck Institute for Heart and Lung Research in Bad Nauheim and Giessen University, a team of scientists under the leadership of Dr. Soni Savai Pullamsetti is uncovering potential reasons why pulmonary arteries become narrowed and lead to pulmonary hypertension. Recent experiments, highlighted in a Nature Medicine journal article, “Pro-proliferative and Inflammatory Signaling Converge on FoxO1 Transcription Factor in Pulmonary Hypertension,” uncovered that a lack of FoxO1 signaling can lead to characteristics of pulmonary hypertension, both in vitro and in vivo.
At the Max Planck Institute for Heart and Lung Research in Bad Nauheim and Giessen University, a team of scientists under the leadership of Dr. Soni Savai Pullamsetti is uncovering potential reasons why pulmonary arteries become narrowed and lead to pulmonary hypertension. Recent experiments, highlighted in a Nature Medicine journal article, “Pro-proliferative and Inflammatory Signaling Converge on FoxO1 Transcription Factor in Pulmonary Hypertension,” uncovered that a lack of FoxO1 signaling can lead to characteristics of pulmonary hypertension, both in vitro and in vivo.
For patients with pulmonary hypertension, this could mean new treatment options that are more efficacious than those already available. “A potential new form of therapy could focus on increasing the activity of FoxO1 in the pulmonary arteries of patients,” said Werner Seeger, who was a co-author on the study, in a news release from the Institute. After increasing FoxO1 signaling in pulmonary vessel walls, “Rats suffering from pulmonary hypertension were essentially cured,” continued Dr. Seeger.
[adrotate group=”4″]
The basis behind why FoxO1 signaling is crucial for preventing pulmonary hypertension is proliferation. In the disease state, pulmonary arterial cells grow and divide at an abnormally rapid rate, leading to narrowed vessels. “The vessel walls of pulmonary arteries are constantly being renewed,” said Dr. Pullamsetti. “A complex interplay of many factors normally ensures that the ratio between dividing and dying cells is balanced.” This interplay seems to be dysfunctional in pulmonary hypertension patients. “In these patients, FoxO1 is not sufficiently active, so that the activity of various genes is not properly controlled,” explained Dr. Pullamsetti.
These findings were made through a comprehensive set of experiments. Along with Drs. Pullamsetti and Seeger, lead author Rajkumar Savai and colleagues analyzed human lung tissues containing pulmonary vessels and pulmonary artery smooth muscle cells from patients with and without pulmonary hypertension. There was a noticeable downregulation of FoxO1 in diseased tissue, as well as an inactivation of FoxO1 via phosphorylation and nuclear exclusion.
To see if they could replicate these results, the team used pharmacological inhibition and genetic ablation of FoxO1 in cell culture. “If we switch off FoxO1 by means of genetic or pharmacological intervention, the vascular wall cells divide more frequently,” said Savai.
[adrotate group=”3″]
Perhaps more importantly, FoxO1 activity could be reconstituted in animals with induced pulmonary hypertension through intravenous or inhaled paclitaxel or gene therapy. These interventions returned control over the cell cycle and transformed the pulmonary artery smooth muscle cells back into a quiescent phenotype.
In the article, the authors attribute the fact that pulmonary hypertension has not yet been cured to the underlying reasons of pulmonary hypertension: proliferative and inflammatory pathways. Since a number of these pathways are controlled by FoxO1 signaling, FoxO1 is an essential player in addressing pulmonary hypertension.



